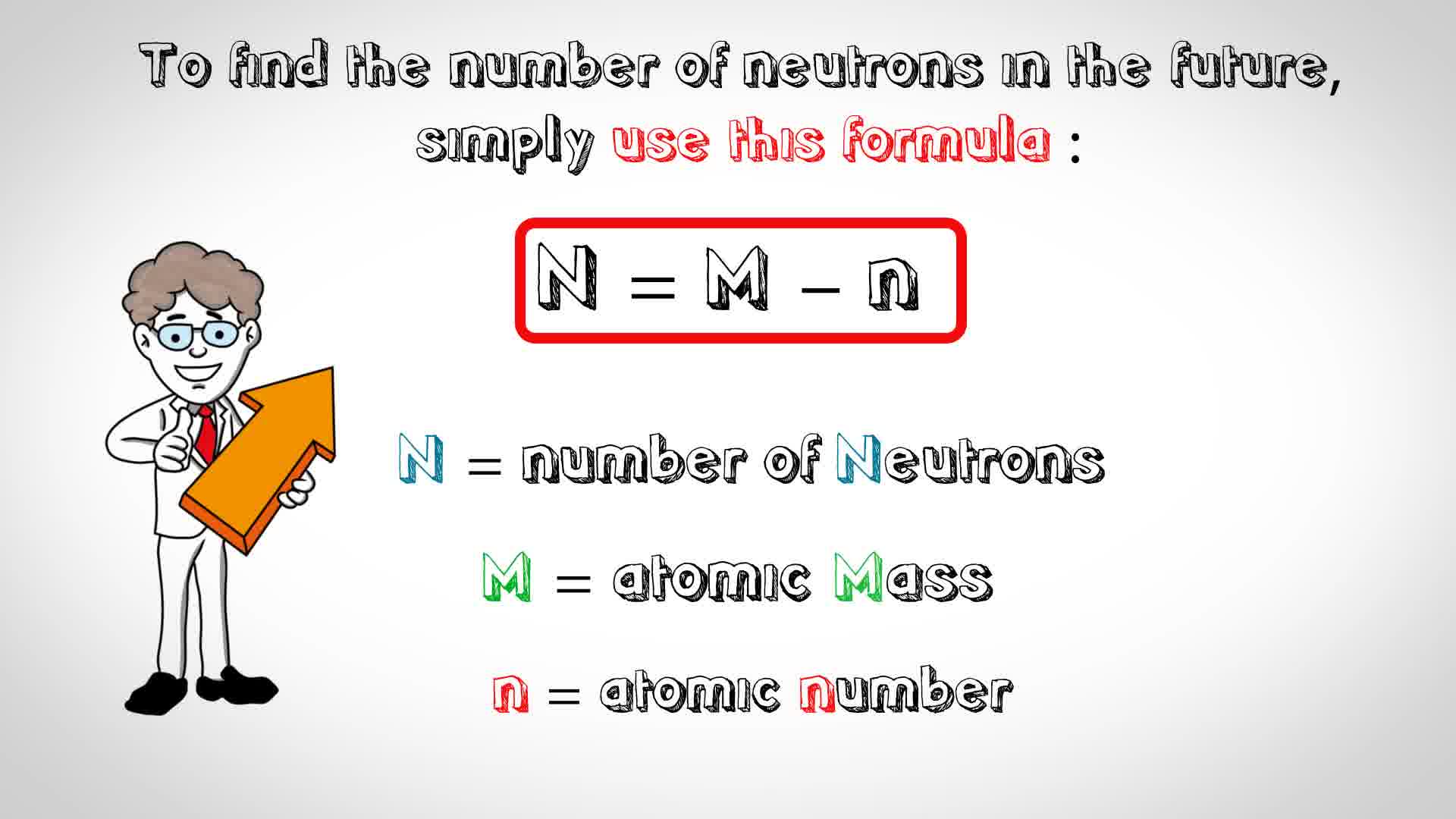Formidable Tips About How To Find Out The Number Of Neutrons

Finding the number of neutrons in a regular atom locate the element on the periodic table.
How to find out the number of neutrons. 6) subtract the atomic quantity from the atomic mass. What is the number of. How to find electrons, when an atom is ionized, it loses one or.
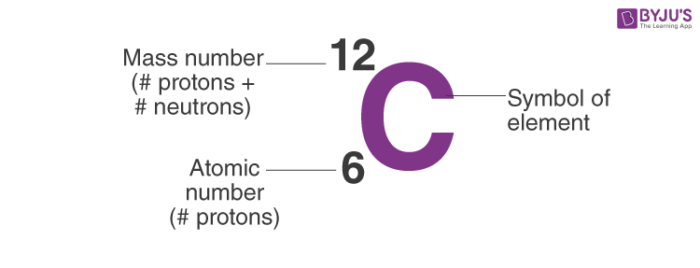
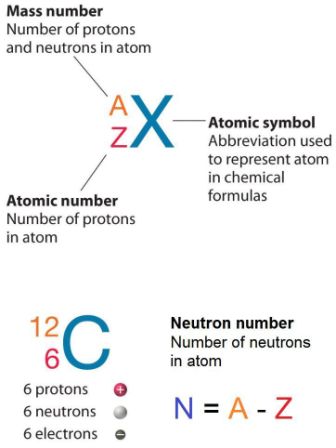
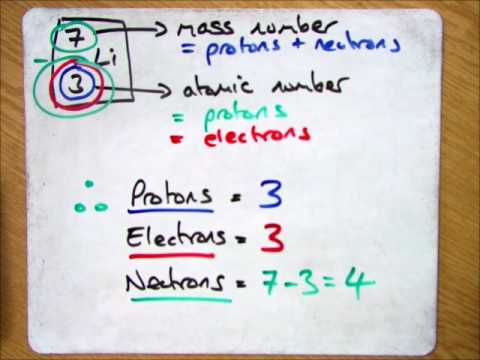
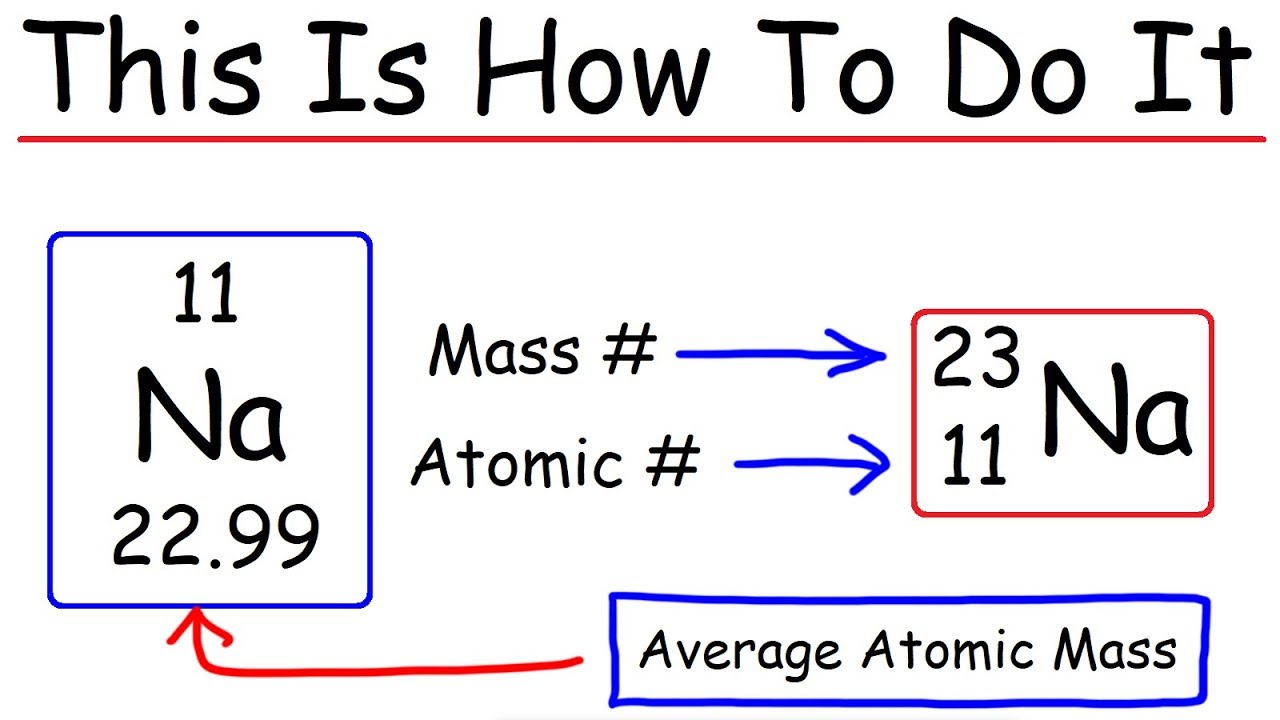
How to calculate the number of neutrons in an atom given the information provided by the periodic table of elements When we write the symbol for an atom, we can place its mass number at the top left and its atomic number at the bottom left. Identify the mass number of the isotope.
Identify the atomic number of the element. How do you find the neutron number? They are both equal thus making the atom have a neutral charge.
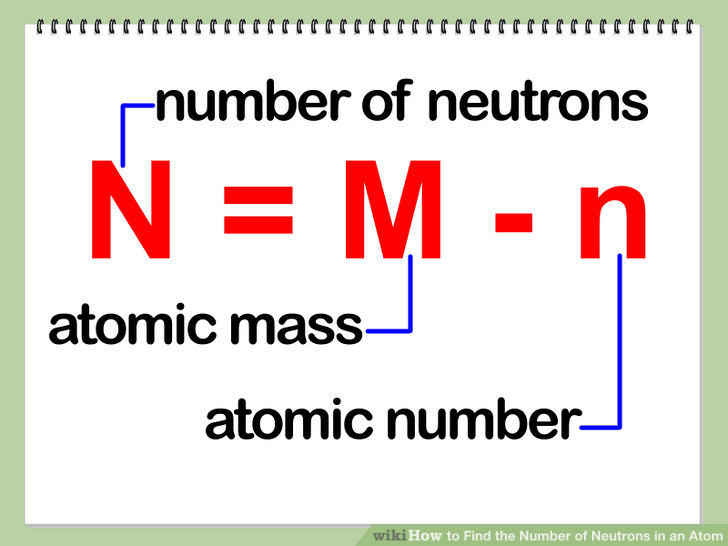
That is, neutron number (n) = atomic mass. Thus, the number of neutrons in an element is obtained from the difference between the number of atomic masses and the number of atoms. The relative atomic mass is the number of.
To calculate the number of neutrons in the nucleus of an atom is simple. Almost all atoms contain three main particles: You take the atomic, or proton number of the element, and you subtract it from the element's mass.
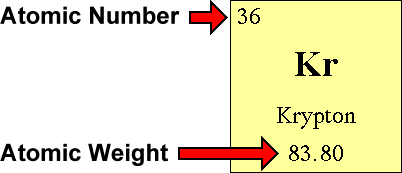
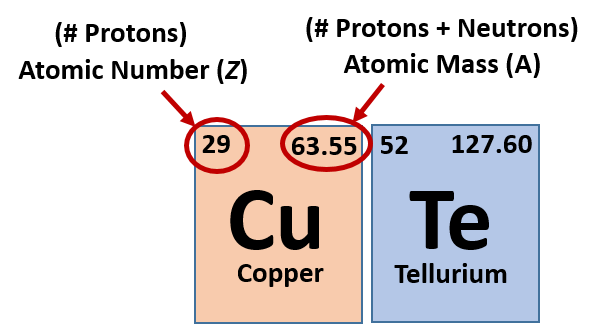
Find the element’s atomic weight. # of protons = atomic number. This chemistry video tutorial explains how to calculate the number of protons, neutrons, and electrons in an atom or in an ion.
The atomic number is the number of protons or electrons. How to find the number of neutrons in an isotope particles in atoms. Ought detect the quantity of neutrons, you will need ought subtract the atomic quantity from the atomic mass.
The number of neutrons can be calculated by simply looking at the periodic table of elements. The number of neutrons in an atom can be figured by subtracting the atomic number from the mass number. Find the element’s atomic number.
It also explains the differe. You can use these numbers to calculate the number of.